- Find A Medical Provider
- Auto Injuries
- Common Injuries
- Medical/Pharmaceutical
- Types of Medical Injuries
- Malpractice Injuries
- Drug and Medical Device Injuries
- Drugs and Devices Linked to Cancer
- Opioid Addiction
- Drugs and Devices Known to Cause Injury
- 3M Combat Arms Earplugs – Hearing Loss
- Accutane
- Aciphex
- Actonel
- Actos
- Adderall and Ritalin
- Advair
- Aldara (Imiquimod)
- Alli
- Ambien
- Amiodarone
- Anzemet
- Aptivus
- Aranesp
- Arava
- Atorvastatin
- Avandia
- Benicar
- Birth Control Medication
- Blood Thinners
- Essure
- Fosamax (Alendronate Sodium)
- Gadolinium-Based MRI Contrast Agents
- Granuflo
- Hernia or Surgical Mesh Injuries
- Hydroxycut
- Inferior Vena Cava Filters
- Invokana Toe and Foot Amputations
- Ketek
- Levaquin
- Lipitor
- Mirapex
- Neurontin
- Onglyza
- Over-the-Counter Medications
- OxyContin
- Paxil
- Power Morcellators
- Pradaxa
- Propecia
- Reglan
- Talc Powder
- Trasylol
- Valsartan
- Viagra
- Xolair
- Zelnorm
- Zoloft
- Work Injuries
- Sports Injuries
- Marketing Services
- Blog
List your practice on InjuredCare | Log in / Sign up
Xolair

Xolair has been formulated and distributed by the biotechnology group Genentech since 2003, when the FDA approved its use to treat asthma. In 2014, the FDA expanded the approved use of Xolair to include treatment of chronic idiopathic urticaria (severe hives). Xolair is not a general antihistamine in that it is not approved or recommended for general allergy symptoms or skin conditions. Genentech and its marketing group, Novartis Pharmaceuticals, have faced substantial scrutiny for Xolair's significant risks and side effects.
Uses for Xolair
Omalizumab helps asthma sufferers by reducing their allergy symptoms. It typically is prescribed for patients who experience chronic and severe symptoms as an add-on to an existing treatment plan. It also is used to treat patients with chronic hives that do not react to other antihistamines.
Xolair is administered as an injection by a qualified physician. Patients who receive Xolair injections are given single or multiple injections once every two to four weeks. Xolair is most effective in children over 12. Children under 12 are more prone to experiencing side effects from Xolair injections.
Severe side effects of Xolair can include:
- Anaphylaxis - a life threatening allergic reaction that can lead to swelling of the mouth or throat, making it nearly impossible to breath. These anaphylactic symptoms closely mimic an asthmatic episode. This makes it harder to identify quickly, increasing the overall risk of this already severe reaction.
- Cancer
- Infection (parasitic)
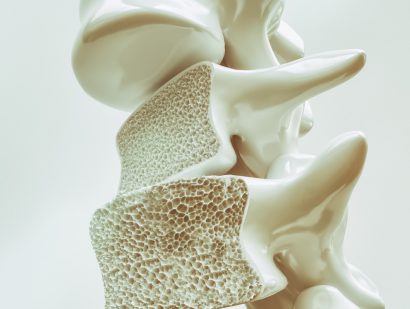
- Bone fractures
- Blood vessel inflammation
- Stroke
- Heart attack
The FDA, Genentech, and Novartis kept patients in the dark
Xolair has improved the health and overall quality of life for many patients. However, there have been multiple lawsuits against Genentech and Novartis for not making the severe side effects clear to patients before use. In 2007, the FDA to required Genentech to place a black label on its bottles warning patients of the potential for anaphylaxis. Two years later, the FDA began to investigate Xolair and its effect on blood vessels in the heart and brain. This launched a five year study on the long-term effects of Xolair and whether it increases risk of experiencing a heart attack or stroke. After multiple reviews of this study, the FDA in 2014 required Genentech to amend the Xolair warning label to include the increased risk of heart attack and stroke.